Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 ,
, -dimethylacrylamide hydrogels
-dimethylacrylamide hydrogelsFukazawa, Tomoko*; Ikeda, Naohiro*; Tabata, Mayu*; Hattori, Masataka*; Aizawa, Mamoru*; Yunoki, Shunji*; Sekine, Yurina
Journal of Polymer Science, Part B; Polymer Physics, 51(13), p.1017 - 1027, 2013/07
Times Cited Count:36 Percentile:75.18(Polymer Science)Ata, Seisuke*; Oka, Toshitaka; He, C.-Q.*; Odaira, Toshiyuki*; Suzuki, Ryoichi*; Ito, Kenji*; Kobayashi, Yoshinori*; Ougizawa, Toshiaki*
Journal of Polymer Science, Part B; Polymer Physics, 48(20), p.2148 - 2153, 2010/10
Times Cited Count:6 Percentile:20.25(Polymer Science)Surface morphology of bisphenol-A polycarbonate (BAPC) thin films with thickness ranging from 30 to 1000 nm on silicon substrates was studied by atomic force microscopy (AFM). The films were prepared by spincasting from 1,2-dichloroethane solutions of 0.25-5.0 wt % BAPC. Even though longer annealing than 250 h was necessary for complete crystallization for bulk BAPC, high crystallinity was observed for 30 nm thick film after annealing at 200  C for 48 h in vacuum. Positron annihilation lifetime spectroscopy (PALS) measurements showed that the free volume hole size in 30 nm thick film was larger than that of bulk at 200
C for 48 h in vacuum. Positron annihilation lifetime spectroscopy (PALS) measurements showed that the free volume hole size in 30 nm thick film was larger than that of bulk at 200  C. Comparison of the BAPC concentration in the precursor solution with the overlap concentration suggests that the high crystallinity of the 30 nm BAPC film is due to less entangled chains caused by rapid removal of the solvent from the dilute solution.
C. Comparison of the BAPC concentration in the precursor solution with the overlap concentration suggests that the high crystallinity of the 30 nm BAPC film is due to less entangled chains caused by rapid removal of the solvent from the dilute solution.
Kim, D.*; Kyu, T.*; Hashimoto, Takeji
Journal of Polymer Science, Part B; Polymer Physics, 44(24), p.3621 - 3630, 2006/12
Times Cited Count:5 Percentile:16.83(Polymer Science)Various topological phase diagrams of blends of main-chain liquid crystalline polymer (MCLCP) and flexible polymer have been established theoretically in the frame-work of Matsuyama-Kato theory by combining Flory-Hussing (FH) free energy for isotropic mixing, Maier-Saupe (MS) free energy for nematic ordering in the constituent MCLCP, and free energy pertaining to polymer chain-rigidity. The calculated phase diagrams exhibit liquidus and solidus lines along with a nematic-isotropic (NI) transition of the constituent MCLCP. Subsequently, thermodynamic parameters estimated from the phase diagrams hitherto established have been employed in the numerical computation to elucidate phase separation dynamics and morphology evolution accompanying thermal-quench induced phase separation of the MCLCP/polymer mixture.
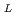 -proline methyl ester) gel membrane in LiCl solution
-proline methyl ester) gel membrane in LiCl solutionChen, J.; Asano, Masaharu; Tsubokawa, Norio*; Maekawa, Yasunari; Yamaki, Tetsuya; Yoshida, Masaru
Journal of Polymer Science, Part B; Polymer Physics, 43(20), p.2843 - 2851, 2005/10
Times Cited Count:0 Percentile:0.01(Polymer Science)Impedance spectra analysis of a thermo-responsive poly(acryloyl-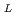 -proline methyl ester) (poly(A-ProOMe)) hydrogel membranes in an aqueous solution of LiCl was carried out using a simple equivalent model. The hydrogel membrane was synthesized by
-proline methyl ester) (poly(A-ProOMe)) hydrogel membranes in an aqueous solution of LiCl was carried out using a simple equivalent model. The hydrogel membrane was synthesized by  -radiation-induced polymerization and crosslinking of A-ProOMe monomer aqueous solution in a glass-cast. By means of the impedance spectra analysis, a novel method for the calculation of the ionic conductivity of the hydrogel membranes in LiCl solution was proposed. The calculated ionic conductivity was in agreement well with the determined value. In addition, effects of temperature and LiCl concentration on the impedance spectra and ionic conductivity of the gel membrane were analysized. Results indicated that the impedance spectra analysis is a very useful tool for evaluating the electric properties of gel membranes in an electrolyte solution. The poly(A-ProOMe) gel membrane in 1.0 M LiCl solution showed a high ionic conductivity of about 0.2 S/cm at 14
-radiation-induced polymerization and crosslinking of A-ProOMe monomer aqueous solution in a glass-cast. By means of the impedance spectra analysis, a novel method for the calculation of the ionic conductivity of the hydrogel membranes in LiCl solution was proposed. The calculated ionic conductivity was in agreement well with the determined value. In addition, effects of temperature and LiCl concentration on the impedance spectra and ionic conductivity of the gel membrane were analysized. Results indicated that the impedance spectra analysis is a very useful tool for evaluating the electric properties of gel membranes in an electrolyte solution. The poly(A-ProOMe) gel membrane in 1.0 M LiCl solution showed a high ionic conductivity of about 0.2 S/cm at 14 C. The temperature-dependence of the ionic conductivity was a complex nonlinear form due to the volume phase transition of the thermo-responsive poly(A-ProOMe) gel membrane, and the volume phase transition temperature appeared to be decreased with the increase in the LiCl concentration.
C. The temperature-dependence of the ionic conductivity was a complex nonlinear form due to the volume phase transition of the thermo-responsive poly(A-ProOMe) gel membrane, and the volume phase transition temperature appeared to be decreased with the increase in the LiCl concentration.
 -isopropylacrylamide)/potassium chloride gel electrolytes
-isopropylacrylamide)/potassium chloride gel electrolytesChen, J.; Maekawa, Yasunari; Yoshida, Masaru; Tsubokawa, Norio*
Journal of Polymer Science, Part B; Polymer Physics, 40, p.134 - 141, 2001/11
Times Cited Count:7 Percentile:27.52(Polymer Science)no abstracts in English